The speed at which the first COVID-19 vaccines were developed was extraordinary. Learn more about US.
Coronavac Wikipedia Bahasa Indonesia Ensiklopedia Bebas
Two mRNA vaccines one by Pfizer and one by Moderna were approved late in 2020.
Covid vaccine wiki. In another study which was published from West Bengal a state in Eastern India has shown that 7727 of people want to take the COVID-19 vaccine. Vaccine acceptance in India. The only evidence on safety of mRNA vaccines comes from small phase I and phase II trials of SARS-CoV-2 vaccines with follow-up typically less than two months.
Comirnaty Pfizer COVID-19 vaccine New Zealand data sheet PDF 12 MB How side effects are identified In the COVID-19 vaccine clinical trials people were followed very closely for side adverse effects for 2 months after the second dose of the vaccine. The synthetic mRNA fragment is a copy of the specific part of the. However this vaccine is an inactivated vaccine with an adjuvant that is routinely used in many other vaccines with a documented good safety profile including in pregnant women.
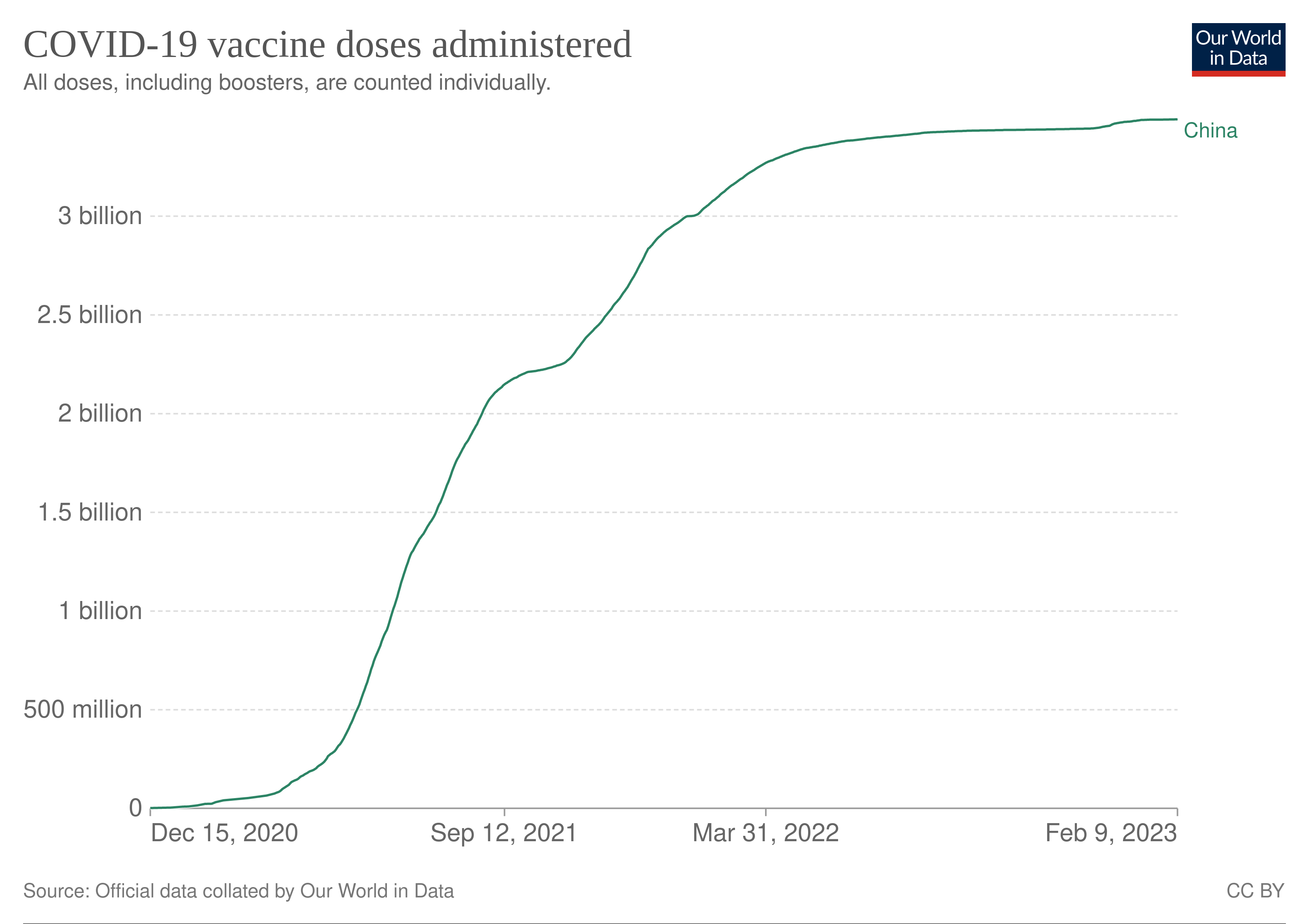
The COVID-19 vaccination program in Canada is an ongoing intergovernmental effort coordinated between the bodies responsible in the Government of Canada to acquire and distribute vaccines to individual provincial and territorial governments who in turn administer approved COVID-19 vaccines during the COVID-19 pandemic in CanadaSome provinces have asked local municipal governments. We have previously looked into the history of vaccine development. It took only 10 years from the discovery of the pathogen to the development of the first vaccine.
Messenger RNA vaccinesalso called mRNA vaccinesare some of the first COVID-19 vaccines authorized for use in the United States. Novavax is a Maryland-based biotechnology firm that is taking a traditional approach to developing a vaccine against the coronavirus disease 2019 COVID-19. COVAX aims to accelerate the development and manufacturing of COVID-19 vaccines and guarantee fair and equitable access for every country.
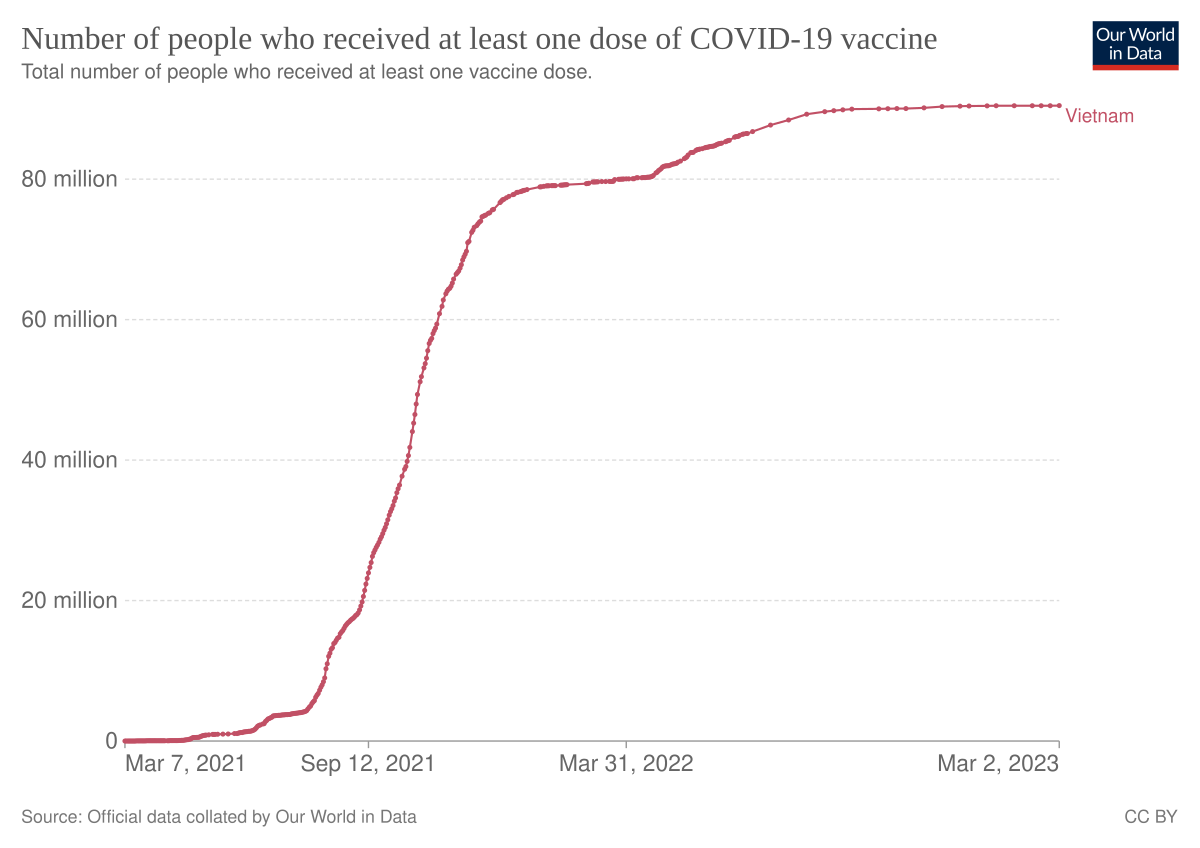
The vaccine is given by intramuscular injection into the deltoid muscle. One study published on the vaccine acceptance is showing that 795 of people from New Delhi a state in Northern India want to take a COVID-19 vaccine. 206 rows Health officials distribute the Moderna COVID-19 vaccine to frontline health workers and.
Early in the development of mRNA vaccine technology fetal cells were used for proof of concept to demonstrate how a cell could take up mRNA and produce the SARS-CoV-2 spike. From Simple English Wikipedia the free encyclopedia A COVID-19 vaccine is any of the vaccines used against the Coronavirus Disease 2019 virus caused by SARS-CoV-2. In July 2020 more than 150 vaccines were being developed in different laboratories.
The Johnson Johnson COVID19 vaccine is used to provide protection against infection by the SARS-CoV-2 virus in order to prevent COVID-19 in people aged eighteen years and older. To trigger an immune response many vaccines put a weakened or inactivated germ into our bodies. Elasomeran codenamed mRNA-1273 and sold under the brand name Spikevax is a COVID-19 vaccine developed by Moderna the United States National Institute of Allergy and Infectious Diseases NIAID and the Biomedical Advanced Research and Development Authority BARDA.
The available data on the COVID-19 vaccine BIBP in pregnant women are insufficient to assess either vaccine efficacy or vaccine-associated risks in pregnancy. New Approach to Vaccines mRNA vaccines are a new type of vaccine to protect against infectious diseases. The measles vaccine was found relatively rapidly.
MRNA vaccines do not affect or reprogram DNA inside the cell. Novavax COVID-19 vaccine. The Moderna COVID19 vaccine pINN.
They compared the results between people who had and hadnt been vaccinated. Another advantage of RNA vaccines is that since the antigens are produced inside the cell they stimulate cellular immunity as well as humoral immunity. As of February 27 2021 large-scale Phase 3 clinical trials are in progress or being planned for two COVID-19 vaccines in the United States.
The company began a Phase 3 trial of its vaccine candidate NVXCoV2373 in the United Kingdom in September and a second Phase 3 trial in the United States and Mexico in December. Moderna designed their mRNA-1273 vaccine for COVID-19 in 2 days. COVID-19 vaccine clinical trials including vaccines in earlier stages of development by visiting clinicaltrialsgov.
The mRNA COVID-19 vaccines produced by Pfizer and Moderna do not require the use of any fetal cell cultures in order to manufacture produce the vaccine. It is authorized for use in people aged 12 years and older in some jurisdictions. COVID-19 qarşı peyvənd.
Severe systemic adverse events were reported by 5 to 10 percent of trial subjects. COVAX is co-led by the Coalition for Epidemic Preparedness Innovations CEPI Gavi and WHO with UNICEF as a key delivery partner and PAHO as the procurement agent in the Americas. Systemic adverse events such as fatigue muscle aches headache and chills are common.
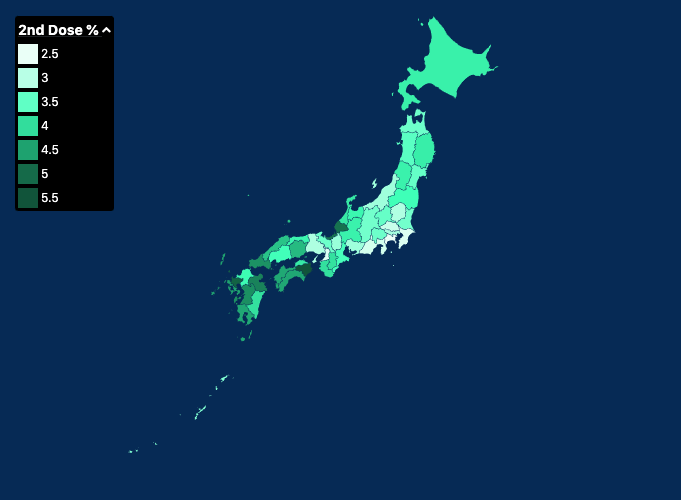
Covid 19 Vaccination In Japan Wikipedia

Vaksin Covid 19 Johnson Johnson Wikipedia Bahasa Indonesia Ensiklopedia Bebas
China S Covid Vaccine From Sinopharm Is 86 Effective Uae Says

Covid 19 Vaccination In Malaysia Wikipedia

Covid 19 Vaccination In Ghana Wikipedia

Covid 19 Vaccine Certification A Contentious Proposal Impact Ethics
How The U S Government Bolstered Moderna S Covid 19 Vaccine Candidate Drug Discovery And Development

Mrna 1273 Wikipedia A Enciclopedia Livre

Covid 19 Vaccination In Nepal Wikipedia

Covishield A Timeline Of The Covid 19 Vaccine Manufactured By Serum Institute Of India Latest News India Hindustan Times

Coronavac Wikipedia Bahasa Indonesia Ensiklopedia Bebas

Covid 19 Vaccination In South Africa Wikipedia